 Photo Credit: PerkinElmer
Photo Credit: PerkinElmer
As vaccination drives kick into high gear, parts of the world are
progressively opening up borders and easing restrictions on events and
travel. It appears that a return to a semblance of normalcy is within
reach. At the same time, recurring waves of COVID-19 stand as reminders
that recovery must be undertaken with caution, and with robust measures
in place.
Testing continues to be critical as the world starts to
think about returning to travel and business events. Still, there are
some lingering misconceptions on the topic. To help bridge information
gaps in the travel community, we speak to the experts at PerkinElmer, a
leading test kit manufacturer and the world’s first provider of
end-to-end SARS CoV-2 testing solutions.
Testing Myths Debunked
Myth #1: With vaccination rates increasing, testing can take a backseat?
Although existing vaccines are able to prevent symptoms, there is increasing evidence that vaccination can only reduce, without fully eliminating, the risk of infection and transmission.
Testing
remains integral to containment efforts and a key part of border entry
requirements for the conceivable future. Almost across the board in
Asia, travellers or returning residents must present negative test
results to enter destinations. This is the case in destinations
including Hong Kong, Singapore, Indonesia, Taiwan, Sri Lanka, Australia,
and so on.
While some countries are easing rules for entry
(e.g. Qatar) and event attendance for vaccinated individuals, the
reality is that people who have received their shots can still be
asymptomatic carriers.
Vaccinated individuals carry low viral loads, making it necessary for tests to have maximum sensitivity to avoid false negatives, PerkinElmer stressed.
A recent publication reported that vaccine efficacy may be compromised when it comes to SARS-CoV-2 variants. Test kits that can detect variants, such as PerkinElmer’s two available kits, will continue to play a central role going forward.
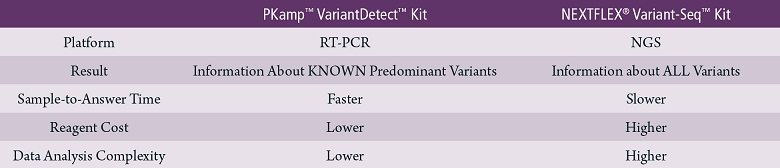
PerkinElmer’s
kits offer two SARS-CoV-2 second-tier assay options, giving labs the
flexibility to implement one or both solutions based on the information
being sought and workflow considerations.
Myth #2: Non-invasive tests are unreliable?
Currently,
the most common ways to obtain test samples are from up the nose or the
back of the throat. While non-invasive methods like spitting into a
tube are available, they are believed to be less accurate.
Not
true, PerkinElmer says. Saliva testing can have a comparably high level
of accuracy as long as the most sensitive PCR tests are used. In fact,
New Zealand’s airport immigration workers are already using PerkinElmer
tests with saliva as a matrix.
These tests designed by PerkinElmer build on the commercially available test kit with the lowest limitation of detection (LOD) in the market according to the U.S Food and Drug Administration (FDA).
The
reason saliva is thought to be a less-than-ideal sample is because its
abundance of impurities can have a negative impact on PCR testing.
PerkinElmer’s chemagen nucleic acid extraction technology overcomes
these challenges, enabling reproducible extraction of high-yield and
high-quality RNA from human saliva.
This alternative method could
be big for travel and event organisers, who may have a difficult time
attracting attendees given the invasiveness of existing pre-event
testing.
 Saliva spit tests can yield comparatively high levels of accuracy if the most sensitive tests are used. Photo Credit: AnnaSivak/Getty Images
Saliva spit tests can yield comparatively high levels of accuracy if the most sensitive tests are used. Photo Credit: AnnaSivak/Getty Images
Myth #3: Mass COVID-19 testing is inefficient and unviable?
Not
only is saliva collection easier on patients, it is also faster and
more scalable for those carrying out the tests, according to
PerkinElmer.
For mass testing needs, PerkinElmer manufactures a
molecular diagnostic test kit approved to screen pooled, asymptomatic
individuals. Pooling samples can result in five times the cost savings,
PerkinElmer shared.
Late last year, the FDA allowed sample
pooling with PerkinElmer’s New Coronavirus Nucleic Acid Detection Kit,
increasing the number of individuals who can be tested, without
increasing demands on resources. If a pooled test is negative, all the
individual samples are considered negative. If it comes back positive,
each of the individual samples in that pool are tested again separately.
Pooling could certainly help address cost and efficiency
concerns, the two biggest challenges in post-pandemic travel management
and pre-event testing.
To simplify the process for event and
travel managers, PerkinElmer — the only test manufacturer that also
provides medtech solutions and end-user delivery — offers the COVID-19 Enterprise Solution and Test Passport encompassing sample collection, testing, reporting results and entry clearance.
The
COVID-19 Enterprise Solution is customisable to different requests,
such as in the case of a concert organiser who had asked for attendee
test results to be reflected in concert tickets for greater efficiency
in clearance.

PerkinElmer’s end-to-end solution for businesses include sample collection, testing, and results delivered to ‘test passports’.
Whether
you are a travel agency, TMC, or event planner, testing will be a big
part of business resumption. The situation is still evolving, and new
information about the virus continues to surface. PerkinElmer, as the
world’s only one-stop provider of testing solutions, can help fill a gap
by combining medical expertise and business solutions, helping
organisations get back to business safely.
For more information, visit https://perkinelmer-appliedgenomics.com/home/sars-cov-2-testing-solutions/covid-passport/.

This article is brought to you by PerkinElmer